THE U.S. E.P.A. is LYING TO YOU!
According to RADICAL environmentalists world-wide, the CO2 produced by the use of fossil fuels is producing enough carbon dioxide "emissions" to create a shift in the global climate, significant enough to cause problems with the environment, melting of the polar ice caps, and eventually, our extinction.
But their so-called "research" is based on flawed logic. Not only does it assume that MAN MADE CO2 is "the problem" it also assumes that CO2 is the ONLY so-called "greenhouse gas" on the planet, AND that man-made CO2 is the primary driving force for "global climate change". Of course, anyone with basic high school chemistry and general science knowledge should be able to see that this is completely ridiculous. Still, for the sake of argument, I'll begin by demonstrating that CO2 levels are NOT causing global warming because of any specific human activity (particularly the kind of activity that burns fossil fuels). Then I shall demonstrate that CO2 is NOT the primary 'greenhouse gas', thus demonstrating that focusing on CO2 is really, really narrow-minded (if not just simply 'clueless'). And finally I'll demonstrate a possible model for showing how and why solar cycles happen, and why we are currently in a 'maximum output' period. And if that's not enough for you, I've even proposed a possible motivation for the "Global Climate Change" hoax, and who it is that would benefit from the use 'Global Warming' for political and/or economic purposes.
Regardless of any estimates you might make, or rectal extrapolations, or outright 'wild freaking guesses'
about the amount of CO2 being put into the atmosphere by the USA, I think
THIS
article pretty much says it all. Note the chart (a little small, still usable) showing the levels of CO2
around the globe. Note where the 'hot spots' are:
FirstNext, I'll bore you with some simple calculations, which you can repeat yourself.
Feel free to report any errors to me so that I can correct them.
I once did an experiment in which I held my breath for approximately 1 minute, and using an exhaust gas analyzer, I measured the percent O2 in my exhaled breath. I discovered that after holding my breath for a minute, the percent oxygen was approximately 10%, less than half of normal (20.9%). I did this to see how efficient my lungs were at extracting oxygen from the air. I found that I could get it as low as 5% if I held my breath long enough, but below that I had some trouble holding my breath. Because I was "at rest" when I did this, it is a good representation of the amount of oxygen that I consume on average, while "at rest".
For sake of calculations, we must assume that every molecule of Oxygen inhaled and metabolized by your body produces one molecule of CO2. By mass ratio, this is 44 grams CO2 per 32 grams O2. Volumetrically, this is 44 grams for every 22.4 liters (1 mole gas at STP) of pure oxygen.
With the assumption that an AVERAGE lung capacity is approximately 1 liter (my lung
capacity is approximately 2 liters, measured while I was in the Navy, in a preliminary
examination I took for working with asbestos), and if we assume that an average person
could hold his/her breath for 1 minute and utilize 50% of the O2 in that breath, at
rest, such that it represents the normal average oxygen consumption by that individual,
we can calculate the total CO2 production as follows:
1 liter air
* (1440 minutes/day)
* (1/22.4 liters/mole)
* (0.209 moles O2/mole air)
* 1/2 (50% utilization)
* 1 mole CO2 per mole O2
* 44 grams / mole CO2
= ~300 grams CO2 per person per day
At 2.2 lbs per KG, that's equal to 0.66 lbs per day.
NOW, using this information, and extrapolating out the entire population of the world, roughly 5 billion people, we have approximately 3.3 billion lbs of CO2 generated per day by people exhaling, or 3.3 billion lbs per day of CO2.
OK, so I admit that my car DOES put out CO2, but it's a LOT better than CO or HC emissions. So what are the wacky environmentalist extremists whining about NOW? Well it seems that THEY think that my car is putting out too MUCH of the same gas that every human on the planet is exhaling right now. So let's see JUST how much CO2 I "pollute" the environment with.
With the assumption that 1 gallon of gasoline (roughly 6 lbs) burns completely, and thereby produces approximately 1 molecule CO2 for every atom of carbon, AND assuming that gasoline is a PURE hydrocarbon compound, with very few double bonds between carbon atoms, the mass of gasoline will be approximately 14 grams for every 12 grams of carbon. Ideal gasoline for '100 octane' measurements is 1,1, dimethyl-hexane, which has 8 carbons, and 18 hydrogens, which is very very close to this ratio. Adding oxygenate may alter this slightly, but in a direction that REDUCES the result of the calculation.
SO, for 1 gallon of gasoline:
1 gallon gasoline
* ~6 lbs per gallon
* 12 g Carbon / 14 g gasoline (grams cancel out)
* 44 g CO2 / 12 g Carbon (grams cancel out again)
= ~19 lbs CO2 per gallon of gasoline.
OK, this is a reasonable figure. From this we can extrapolate that, if every vehicle
in the world (estimate 500 million, 1 per 10 people on the planet) were to consume an
average of 1 gallon of gasoline per DAY, [an obvious OVER-estimate, even for the U.S.A.
because it results in 10,000 miles per year with an overall average of 27MPG per car]
then the total production of CO2 would be:
500 million * 19 lbs = 9.5 billion lbs CO2 per day.
For the sake of comparison, this is approximately 3 times the amount breathed by humans AT REST. Assuming that all of these cars are being driven this much, the amount of CO2 "exhaled" by cars is three times that of all of the humans on the planet.
OK, let's assume that this IS important. What does the U.S. EPA say about it? Well, for
starters, you can see for yourself at the following web site:
http://www.epa.gov/otaq/climate/ghgreport/hwy_veh.pdf
According to this document, automobiles in the United States are producing 1,455 Tg of
alleged "greenhouse gasses" per year.
The number stated in Table 2-1 is 1,371.2 Tg (tera-grams, or 10^12 grams), equivalent to
1.37 x 10^12 KG CO2 per year
- or -
3 x 10^12 lbs of CO2 per year (do the math if you don't believe me)
based on calculations for the year 2000.
Per day, this would be ~8 x 10^9 (8 billion) lbs of CO2 from cars in the United States alone.
Based upon THIS figure, how many gallons is that? With 19 lbs of CO2 per gallon, you can easily
calculate that to be 420 million gallons per DAY. That's 420 million. >1 gallon of gasoline
per day for every person in the United States. Now, that assumes that people of "driving age"
that aren't retired, or disabled [roughly half the population], are driving an AVERAGE of 20,000 miles
per year, each. EACH! For a 5 day work week, this would be an average commute of between 70 and 75 miles
(round trip - 35 miles each way, probably over an hours' driving in traffic each way) for every
'working person', plus additional miles for trips to the store, trips to the soccer field, and so on.
And most of the cars people commute in have reasonable 'traffic mileage', with very few S.U.V.'s and
trucks, because at $1.50+ per gallon, people just can't afford to pay >$50 per week for gasoline for
each car that is used for commuting.
And, using these figures, for a 5 year period, the "life" of a car of 100,000 miles would quickly
be exceeded, and that would scare the financial companies something fierce. At best, I would think it
would be less than HALF of that.
Needless to say it seems to me that an exaggeration of facts has taken place by the EPA, and I'd like to see where they get THEIR numbers from.
But chemical equilibrium isn't the ONLY thing keeping the CO2 levels stable. There is also BIOLOGICAL EQUILIBRIUM. Plants will grow FASTER if you increase the CO2 concentration, and proportionally will DEPLETE THE CO2 FASTER whenever there is MORE OF IT in the atmosphere. If you don't believe me, you might want to read about that sort of thing HERE. Suffice it to say that every farmer on the planet should be VERY happy if the CO2 concentrations are being increased.
BUT, as we know, the wacky environmentalist extremists would have us think that even a SMALL change in the amount of CO2 produced by car exhausts (and power plants and anything else that uses fossil fuel) would have a LARGE change on atmospheric concentrations, and would continue to RISE INDEFINITELY without the depletion rate being affected by the higher CO2 concentration, and thus it would take "hundreds of years" to get back to 'normal levels'. SOME warmists think it will take even longer! But the truth is more reasonable. Volcanic activity (which does increase CO2 levels) has a FAR greater effect than all of the cars and power plants combined, because volcanic activity recycles the sedimentary carbonates back into the ocean, and the warmer water in the vicinity of the volcano causes it to 'give up' some of the precipitate CO2 as 'effervescence'. It's a bit like when a soda goes flat as it gets warm. But were it NOT for the volcanic activity under the oceans, the CO2 levels would be LOWER THAN THEY ARE NOW, due to the CONSTANT formation of carbonate as precipitates, and there wouldn't be NEARLY enough CO2 for all of the plants. We should all be VERY happy that the underwater volcanos are making enough CO2, or we'd all starve to death.
Basic chemistry shows that the Hydrogen Carbonate and Carbonate ions (formed by CO2 reacting with water) are both relatively insoluble with MOST metal ions, except for Group I metals. Solubility forms an equilibrium in which SOME of the ion concentration (below the equilibrium constant) will remain in the solution. For more information on carbonates, you can go HERE, but be careful to avoid any 'warmist propoganda' it might lead you to.
So what exactly WOULD happen if underwater volcanos stopped erupting?
A decrease in underwater volcanic activity would cause more carbonate residue to form on the ocean floor.
As rain hydrolizes carbon dioxide from the atmosphere, it deposits it into bodies of water (like the ocean).
Carbonate precipitate then forms, causing the concentration of oceanic carbon dioxide to drop. Since
the amount of dissolved gas that can be at equilibrium with the ocean is a constant based on temperature,
less carbon dioxide is released back into the atmosphere (as part of that equilibrium), and so the carbon
dioxide becomes 'trapped' in the ocean as carbonates. Underwater volcanos, however, would stir up (and warm)
this precipitate, and cause ADDITIONAL carbon dioxide to be released into the atmosphere. A reduction
in the volcanic activity would therefore REDUCE the carbon dioxide naturally released back into the
atmosphere from the ocean (at equilibrium).
Then, as the atmospheric carbon dioxide levels drop, less carbon dioxide is depleted by the rain.
As the rain dumps less carbon dioxide into the ocean, oceanic carbon dioxide levels drop, causing less
precipitation of carbonate residue, and a new (lower) equilibrium concentration of dissolved carbon dioxide
is established in sea water.
As a result of the lower carbon dioxide levels, algea growth diminishes somewhat, producing less oxygen.
Similarly, plant growth on the earth's surface would be reduced slightly, also producing less oxygen.
A shift in worldwide oxygen production would cause an effective increase in carbon dioxide levels in
the ocean from animal respiration, and to some extent, in the atmosphere. You can add man-made sources
to the production if you like. The fact remains that depletion rate is now lower due to the lower concentration
of carbon dioxide.
Eventually, a new equilibrium level is reached where carbon dioxide levels in the atmosphere may be immeasureably
lower than they were before, but not significant enough to cause global warming/cooling. Carbon dioxide
levels in the ocean would be measurably lower, but total dissolved gas concentration would remain roughly the
same.
Oceanic animals breathing the slightly higher concentrations of oxygen may actually flourish a bit more than usual,
which would actually cause an increase in the production of CO2. A new "biological equilibrium" would be
reached in the oceans.
And despite what anybody wants to admit in the wacky environmentalist camp, the same kind of analysis applies when CO2 levels are INCREASED due to car exhaust and power plants and human activity in general, or horse and composting activity (if we abandon cars and go 'natural' in our farming), or there is an increase in the population of all of the animals on the planet for that matter. Whatever the cause, an increase in production rate of carbon dioxide WILL result in all major depletion factors SHIFTING IN THE DIRECTION of DEPLETING THE CARBON DIOXIDE FASTER, no matter HOW MUCH the warmists claim otherwise.
Of course, if our world's carbon dioxide cycle were UNSTABLE, and not at equilibrium, even a slight change (such as an increase over time of the population of humans and animals, or a gigantic forest fire) would have had a DRASTIC effect on our global climate, forcing us to radically bounce from ice age to searing heat and drought, flooding of the coastal lands by repeated cycles of melted polar caps, and all of the other disastrous climactic images you can think of. But we know that the earth's climate has been "at equilibrium" since the previous ice age, which was probably caused by an asteroid or comet hitting the earth. It's something we can NOW prevent ourselves, given enough warning.
As I find more interesting web sites I'll add them here.
Someone honestly wrote an e-mail to me, and I believe it epitomizes what most people on the 'global warming' side of the argument believe. Rather than criticize it point by point (I don't really need to make people into targets like that), I'll simply quote it 'as-is' and summarize my response.
Your argument regarding global warming is somewhat flawed. While humans do exhale allot (sic) of CO2, all of that CO2 comes from the carbon in simple sugars and carbohydrates in the food we eat. All the of the food we eat comes from either plants, or animals that eat plants (or animals that eat animals that eat plats etc...). Plants make their carbon products (sugars, carbohydrates etc...) from C02 in the atmosphere. Thus, all of the CO2 we exhale originally came from plants and all of that carbon originally came from the atmosphere itself. Therefore it is impossible for us to contribute to the over all CO2 accumulation in the atmosphere because all of the CO2 we breath back into the atmosphere was originally take out by plants. Because of this the net increase of CO2 contributed by humans always comes out to zero. It is possible however for gasoline, coal and oil to contribute to CO2 accumulation. All of the carbon that is released when they burn also originally came from plants however it has accumulated over millions of years as opposed to the sort time in which it takes to grow our food. Oil and coal are made of organic matter (plants from millions of years ago. There is allot of carbon trapped in this from as it has had a very, very long time to accumulate. When we burn oil and coal we release this trapped carbon in the form of CO2. Because oil is formed at such a slow rate and we are burning it at such a fast rate in comparison, there is a net increase of carbon going into the atmosphere. Thus while we cannot contribute to CO2 accumulation by exhaling CO2 (because it is the same amount of CO2 that is being taken up by plants that are currently alive) we can contribute CO2 to the atmosphere by releasing it from stored forms such as oil and coal because that amount of CO2 will not be taken up. In short, all of the CO2 that we breath out will become plants again within our lifetime, but none of the CO2 released by burning oil, coal and gasoline will become oil coal and gasoline within our lifetime. Please look into all of this yourself, I'm not trying to deceive you. Misinformation can be very dangerous, as it leads people to ignore problems that are very real. I hope this is enough to change your mind about atmospheric CO2 contribution by humans and at least modify your page in that regard.
Basically, it condescendingly follows the oversimplified assumption that the depletion rate of CO2 remains constant, regardless of changes in the production rate. Yet it can easily be demonstrated that very SMALL concentration changes of CO2 in the atmosphere will cause a significantly HIGHER amount of CO2 to be depleted by rain, due to the equilibrium constant for CO2 forming carbonic acid in rainwater. A small change in either side of the equation (depletion rate or production rate) results in a shift towards the equilibrium concentration of CO2. Following this, CO2 is then depleted within the ocean by precipitation of carbonates.
Also keep in mind that MOST of the world's CO2 already exists in the ocean, AND if there were no undersea volcanos, nearly ALL of the world's CO2 would be trapped there. Instead, undersea volcanos stir up the carbonates, and effervesce the CO2 back into the atmosphere. So a much larger factor on CO2 concentration in the atmosphere (as well as any 'global warming' effects it might cause) would be the presence (or absence) of undersea volcanos. The different in magnitude of scale should have people more concerned about volcanos, rather than the burning of fossil fuels. And there has been a little more volcanic activity recently, off the coast of California.
Thinking of VOLCANIC ACTIVITY, check out the activity on Antarctica! When warmists claim that ice sheets are SLIDING INTO THE OCEAN because of CO2 warming the planet, consider what the VOLCANOS might be causing, and THAT INCLUDES WARMER CLIMATES AROUND THE ANTARCTIC CONTINENT! Remember, a SINGLE VOLCANO can do FAR MORE than ANYTHING that humans are capable of doing when it comes to warming, greenhouse gasses, and overall destruction. Anyone familiar with Mt. Pinatubo or Mt. St. Helens or (gasp) Mt. VESUVIUS (as in the destruction of Pompeii way back in history) should realize this, without any possible argument whatsoever.
As it turns out, CO2 (which has a very SMALL concentration in the earth's atmosphere) is NOT the 'biggest greenhouse gas on the planet'. Strangely coincidental with what I believe is the agenda of the 'global warming' crowd (to restrict the use of energy by the common folk and reserve it only for the elite) the minor effect of CO2 has the hyperfocus microscope trained upon it, while simultaneously ignoring the 'elephant sized' contributor known as WATER, something that human beings aren't affecting in the least.
Any grade-school aged child above the age of 9 understands the basic hydrological cycle that takes place on our planet, and has been taking place for a few million years (since the 'big cool down'). But as Jeff Foxworthy has pointed out from time to time, the average adult is probably dumber than the smartest 10 year old when it comes to science (and just about everything else). Do I really need to explain to everyone how water evaporates, forms clouds, and rains back to earth? Well, I did make reference to it earlier, as the major depletion factor for atmospheric CO2. I had hoped that everyone would understand this kind of common sense.
So here we are talking about water in the atmosphere, something that we ALL know is there, especially
on hot sticky summer days in the S.E. United States and various tropical regions throughout the world.
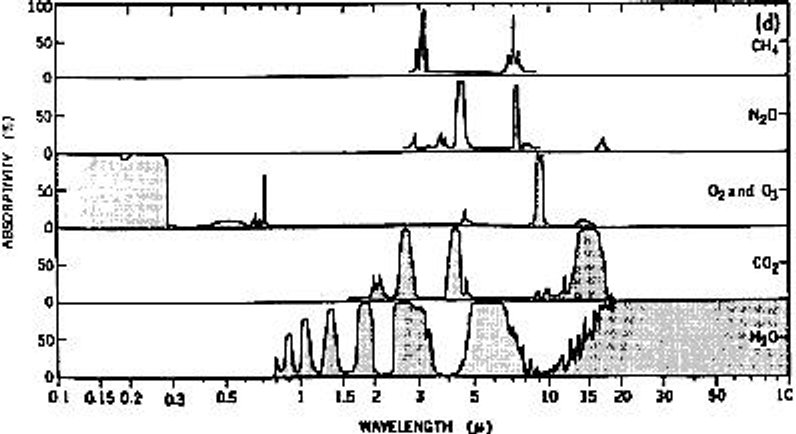
Water absorbs FAR MORE EM RADIATION than does CO2, and the following chart (I believe) demonstrates
this more accurately than I could otherwise describe:

Now I know that someone's going to point out that there is 'complete absorption' of all infrared radiation by CO2 above 13 microns, and I do not disagree with this at all. But you should keep in mind that 13 microns corresponds with a temperature that's LOWER than anything found on Earth, with the exception of Chicago or Alaska or Antarctica at certain times during the winter. The formula of the PEAK IR wavelength (in microns) to temperature (in degrees K) is about 2900 / wavelength. For 13 microns, this would be about 223 deg K or -50 deg C, or -58 deg F. So next time the temperature gets down THAT far, maybe in Alaska or Chicago or Antarctica, you can thank all of that CO2 that it doesn't get any freaking COLDER. But keeping all of the heat in is OBVIOUSLY the job of WATER and OXYGEN, which traps all of the higher energy photons, and not just the wimpy ones that are trapped by CO2. Like I said, CO2 does absorb IR. It's just that H2O absorbs WAY MORE, and is FAR MORE SIGNIFICANT than CO2.
And I would call THAT "the smoking gun" as far as the debate goes. Oh, yeah, it's worth pointing out that the above chart wasn't the ONLY reference I found on this subject. It was merely the one that best illustrated my point. One nice explanation of CO2 absorption lines can be found HERE.
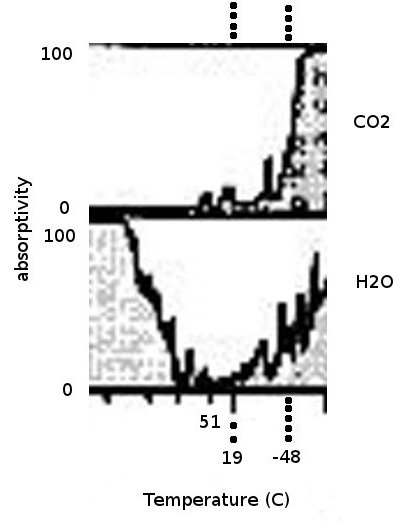
On the right you can see a closeup of the CO2 and H2O portions of the above chart. Zooming in you'll notice that CO2's peak is just BELOW -48 C, which is (again) -58 F. The '10 micron' line corresponds to 17 C, or about 63 F. What can clearly be seen here is the amount of IR energy that is absorbed by water at temperatures above freezing (I would expect water vapor concentrations to be VERY LOW below freezing) with a small 'peak' right around the freezing point. Considering that there is typically several HUNDRED times as much water in the atmosphere as there is CO2, it becomes pretty obvious that the absorption of IR energy by CO2 is INSIGNIFICANT when compared to the absorption of IR energy by WATER with IR energy corresponding to temperatures that are WITHIN THE TYPICAL TEMPERATURE RANGE OF THE PLANET. Now, you might argue that IR emissions include a wide spectrum of energies, and you would be right. But when you look at how much the temperature will change when IR of a particular energy is absorbed, it's pretty obvious that HIGHER ENERGIES (shorter wavelengths) matter MORE than LOWER ENERGIES (longer wavelengths). And, the energy per photon is ALSO higher for shorter wavelengths. The probability that a photon will be absorbed is a function of its energy and the 'macroscopic cross section for absorption' of the material. That last nine-dollar phrase is a function of the 'microscopic cross section' (a known factor) and the concentration times the physical distance through which the photon must pass. Think of it as a marble going through a circle with either LOTS of marbles in it (high probability of interaction) or only 1 marble in it (low probability of interaction). So from these charts, the RELATIVE ABSORPTION of a particular energy of photon will not only depend on its energy (for that specific gas) but ALSO ITS CONCENTRATION WITHIN THE ATMOSPHERE.
Extending this, consider that water can be up to 2 percent of the atmosphere, particularly in hot, humid areas. CO2, however, is typically only 0.04% of the atmosphere. If the average amount of water in the atmosphere were only 0.4% (relatively dry throughout the world, if you compare it against THIS chart), it's still 10 times the concentration of CO2, and can be as much as 50 times the concentration of CO2. THEN, compare the total amount of RELEVANT IR ENERGY that water absorbs as compared to CO2, and it's pretty obvious which one is the "better" greenhouse gas. And, consider that cloud cover during the day can drop temperature by 10's of degrees F, and cloud cover at night can RAISE it by a similarly high amount (though not QUITE as much as the daytime effect). It's OBVIOUS that WATER makes a HUGE DIFFERENCE on the climate. And it's OBVIOUS that the MINUTE levels of CO2 being put into the atmosphere by human activity, by comparison, is NOTHING. NOTHING. It's like, drop in a bucket, which is (basically) NOTHING AT ALL. ZERO, ZIP, NADA. UNMEASURABLE. NO EFFECT. A fly on the scale when you weigh yourself. NO difference.
I have seen at least ONE source attempt to discredit the idea that water vapor has many times the effect of CO2 on global climate. They allude that some kind of 'cycle' exists that somehow amplifies changes in CO2 using water. They are so full of themselves that it is pathetic. Water ABSORBS CO2 FROM THE ATMOSPHERE. This is a well-known chemical reaction. Have you ever heard of ACID RAIN? Do you know where it comes from? That's right, GASES in the atmosphere, like CO2, NO2, NO3, SO2, SO3, etc. are absorbed by the rain, and are DEPLETED FROM THE ATMOSPHERE. We know what the equilibrium constants are for water and for CO2. Spraying water into pure CO2 will CREATE A VACUUM as the CO2 is absorbed. If the pool of water that the spray falls into contains Mg or Ca, the CO2 will PRECIPITATE OUT as MgCO3 and CaCO3, otherwise known as "scale". You can see it on your shower head and bathroom faucets and inside of an old water heater. It is also how shellfish create their shells. Yeah, they bubble CO2 through the water and the hard part of the shell forms. Amazing, isn't it? I learned this when I was in 2nd grade, when we all used straws to blow air through sea water in a glass. You can try THAT experiment, too. So anyone who TRIES to say that the CO2 will 'stay in the atmosphere for hundreds of years' and 'only geological stuff will remove it' needs to get a freaking clue. CO2 is AT EQUILIBRIUM. It is EASY TO DEMONSTRATE with SIMPLE EXPERIMENTS. ADDING TONS OF MORE CO2 to the atmosphere will SIMPLY PRECIPITATE IT ALL OUT IN THE OCEAN. 'Nuff said.
One area of science that's clearly being ignored by the vast majority of those involved in the debate of whether or not 'Global Warming' is just a natural cycle is ASTRONOMY. But why Astronomy, you ask? It's because of the tidal forces on the sun as caused by the planets and their combined effect on solar output.
Recently scientists have ADMITTED that, although sunpsot actiivity has been low, SOLAR OUTPUT in the frequency ranges that matter (i.e. visible light and near infrared) have actually INCREASED, which have then significanlty 'warmed' our earth at a rate above normal. An article at The Register goes into more detail, but instead ADMITS that Mr. Sun may be the cause of the warming trend, and NOT man-made carbon dioxide (as was previously assumed). It also demonstrates how very little many scientists really know about how the sun works.
Let us not forget that Mr. Sun is also a bozillian times more powerful than we humans are. I'd put Mr. Sun in the 900 billion pound gorilla category any day, as compared to anything HUMANS could accomplish. So when Mr. Sun gets hot under the collar, so do we. No questions there, except maybe WHY this happens. And I think I have a pretty good idea what might be the cause.
Astronomers have recently discovered a large number of planets orbiting stars because of the doppler

Our solar system has two very large planets, and about every 60 years they'll share the same orbital
angle to the sun. It is my presumption that the combined gravitational effects of Jupiter and Saturn cause
a significant tidal force on the sun, and this results in a period of time in which solar activity at
certain relative orbital angles from Jupiter and Saturn will be higher than normal. This can either
cause warmer or cooler weather, depending upon the earth's relative orbital position and season.
Tidal effects on earth have been known for millenia, and are easily predicted by the sun's and moon's
Similarly, the combined gravitational force of Jupiter, Saturn, and possibly other planets (Venus, Mars, and Mercury, specifically) will cause 'tidal bulges' to appear on the sun. The next question is obvious: Do the tidal bulges on the sun affect solar output? I say YES!
The probability of a nuclear reaction taking place is the result of several physics parameters, which include the materials involved, their energy level (or 'excitation state'), and the DENSITY of the reactants. In the case of nuclear fission we know that a higher density of fissionable material results in a higher 'reactivity' which allows reactors to be physically smaller and still generate power. In the case of hydrogen fusion (the sun's nuclear reaction) we know that high temperature and pressure are necessary to sustain the reaction. We also know that the sun's internal reactor stabilizes itself between gravity and the outward expansion caused by the fusion reaction in the sun's core. As gravity presses reactants together, reaction rate increases, causing the sun to expand, which lowers the reaction rate. This creates the nice stable nuclear fusion reaction that keeps the sun 'burning' and not exploding.
We also know that there are natural variations in the reaction rate, variations which follow measurable periods in time. Not so amazingly, the sunspot cycle is also an indicator of solar output and follows (interestingly enough) an 11 year period, which is close to the orbital period of Jupiter (11.87 years). However, Jupiter alone is probably not the culprit here; it's most likely a combination of planets that is the key to the sunspot cycle. And since inner planets move FASTER, the time between 'relative positions' lining up is going to be slightly less than 11.87 years. In fact, Jupiter and Mars will line up about every 2 years, and Earth and Jupiter about once a year, and both Venus and Mercury more often still. My proposal is that certain positions of Jupiter's orbit, particularly its apogee and perigee, along with the positions of other planets, may have a direct effect on solar output, as evidenced by sunspot cycles.
Should this, in fact, be the case, you should see a LOT of evidence that compares solar cycles with planetary
positions, especially the apogee and perigee position of Jupiter, and the relative positions of other planets
creating tidal bulges on the sun. And it may be that when the tidal effects are the greatest, you have a
maximum output. In early May of 2000 there was a major planetary alignment (5 planets + moon as viewed from
earth, within 26 degrees of one another) and solar output was at an unusually high maximum as compared to the
sunspot level (which was lower than usual during the 11 year sunspot cycle). A nice graphic is available on
wikipedia HERE. Other planetary alignments
have been taking place over the last several years, in what appears to be a much higher frequency than at other
times, and it will continue (supposedly there's one in 2012, but I couldn't find it with celestia) for a few more
years until the planets happily go about their merry 'unaligned' way. And because of this NATURAL CYCLE, the
solar output is higher than usual, and will remain higher than usual for several more years.
I used 'celestia' to snapshot a few planetary alignments. These show interesting alignments that are about 10 years apart.
May 10, 2000
January 25, 2010 July 2, 2020
Now we can all expect that those in the 'global warming' crowd are going to want to TAKE CREDIT for the obvious cooling pattern that WILL BEGIN within a decade or so, due (as I believe) to the lower frequency of planetary alignments and their relative arrangement to the Earth's apogee and perigee. Those environmentalist wack-jobs (aka man-made global warming fascists) WANT to have all of their ridiculous legislation passed AND ENFORCED for the simple reason that they will want to USE THIS AS "EVIDENCE" that their policied "WORKED" (when in fact it is clearly obvious to those of us with open enough minds that natural cycles are the only thing changing global temperature). So keep in mind that it's 2010 right now when I'm writing this. If what I say is true, you know I said it before it happened, and I used SCIENCE to predict the outcome! Sure, it's a theory, and this is my experiment, to prove or disprove it. We'll just wait and see.
You ARE encouraged to READ THIS FOR YOURSELF.
After much thinking about this issue, I have often considered the possibility that the entire 'Man Made Global Warming' issue exists NOT because the people behind the scenes actually BELIEVE in this pseudo-science, but rather are attempting to bring about some kind of change. Either society itself, or the world's political/economic state isn't what they envision it to be, and they fabricated the 'Man Made Global Warming' issue as a kind of crowbar to throw into the world economic engine in order to make it all happen their way.
This makes a LOT of sense if you read between the lines. And there are a number of people who benefit from this kind of thing. There are many good reasons to make changes, and many sinister reasons as well. But it's still the kind of 'manipulation' that only the fearful would do. And above all other reasons, it is most likely FEAR that drives it.
The Kyoto Protocol was an obvious ploy into forcing the United States to stop being the economic leader that it has been since World War II. Unfortunately many policies have apparently been enacted to this end, and our current situation as an economic leader is rather tenuous at the moment, all thanks to government policies. There are people out there that believe that LOWERING the United States' economic influence will somehow ELEVATE everyone else. What they fail to see of course is that a rising tide DOES lift ALL boats. They see economics as a zero-sum game where one person 'having' means someone else must 'have not'. But of course this MARXIST idea is just NOT true. Flowing money makes everyone who touches it better off. Oppressing one country, like the United States, will cause WORLDWIDE lowering of economic status, because it causes STAGNATION of the flow of money, and eventually results in a worldwide economic recession (or even depression).
Still, there may be OTHER politically based motives, which may actually seem helpful. Right now the entire world is depending upon oil that is being pumped out of the Middle East. And PARTIALLY as a result of all of the money flowing into this region, we have well-funded terrorists committing 'crimes of terror' all over the world. Using 'Man Made Global Warming' as a crowbar into the current energy usage patterns of industrialized nations, you COULD force the invention of other sources of energy (such as hydrogen fusion) as well as the development of electric vehicles that would effectively SHUT OFF THE FLOW OF MONEY to this region of the world. It might be a way to combat terrorism! But if this is the case, WHY LIE ABOUT IT? Lying about something in order to do it 'for our own good' is blatently dishonest, and the KIND of MANIPULATIVE SINISTER BEHAVIOR that 'those in power' have often exhibited for centuries. They deny the fact that people are basically intelligent and tend to make CORRECT choices whenever they are presented with enough information to become INFORMED. Instead, these people whip up frenzy using EMOTION and never really solve anything. Such people must think that everyone else (but them) are idiots.
The bottom line is that WHOEVER is behind the 'Man Made Global Warming' facade should simply come forward and state what the agenda REALLY is. If these people truly believe that the results of their agenda is better for everyone in the world, let them address the United Nations and plead their case, and allow questions and commentary and debate to follow. But using EMOTION to displace LOGIC, and 'whipping people up into a frenzy' to cause mass hysteria to drive their agenda forward, is the kind of SINISTER MANIPULATIVE EVIL CORRUPT OVERLORDING that all too often characterizes evil leaders throughout history.
Lowering down those who have climbed highest up the ladder of success can only benefit those who are at the very top, and those who are at the very bottom. Those in the middle who are doing their best to get ahead find themselves frustrated by obstacle after obstacle. Having someone tell you that you can NOT climb past a certain rung, and you will be PUNISHED if you try, is only going to create a kind of 'clog' on the ladder. But those at the very bottom (who choose NOT to climb) and those ALREADY at the top, will benefit in several ways, not the least of which is that NOBODY ELSE will reach the top (meaning those already there have MORE power and influence and retain it indefinitely), and those at the bottom get to reap the 'freebies' (wealth re-distribution) that elevates them just enough to keep them from rioting. Still, the ones at the very top benefit the most. They're keeping 'the rest of us' from taking over their exclusive little 'country club', after all.
Those who are ALREADY AT THE TOP are sometimes called 'Old Money'. Yes, they're SNOBS. They're
like an economic NOBILITY of sorts, and they don't want to lose their inherited power. Those
at the bottom are sometimes 'the unfortunate', but usually 'the lazy'. And they're often used
by 'Old Money' to further their agenda. It makes you want to feel sorry for those at the bottom,
but giving them MORE helps nobody. Freedom helps EVERYBODY. That's what we need to give them:
MORE FREEDOM. And the 'Man Made Global Warming' agenda is anything BUT freedom.
And I go into a lot MORE HERE. And HERE.
Recently I experimented with a 'robotic' movie maker, with what I might call 'ok' results. Originally it was a simple response to a much shorter work (done by someone else) that attempted to make the 'Tea Party' people look like vacuous idiots. I (and others) thought that the concept of 'robot' movies based entirely on text input were good, but the politics were questionable at best. So I decided to make one of my own, having some spare time on Christmas Eve, 2010.
I had hoped that it would be a somewhat 'darkly comedic' look at what life would be like in
2014 if the Tea Party elections of 2010 fail to cause reform, and if Obama gets re-elected
in 2012 and THEN gets what he REALLY wants.
You can view it HERE (flash video).
Again, THINK FOR YOURSELF! Please!!!